Shop for Plans
Shop for your own coverage
Plans through your employer
Learn about the medical, dental, pharmacy, behavioral, and voluntary benefits your employer may offer.
Learn
Living or working abroad?
Childhood Acute Lymphoblastic Leukemia Treatment (PDQ®): Treatment - Patient Information [NCI]
General Information About Childhood Acute Lymphoblastic Leukemia
Childhood acute lymphoblastic leukemia (ALL) is a type of cancer in which the bone marrow makes too many immature lymphocytes (a type of white blood cell).
Childhood acute lymphoblastic leukemia (also called ALL or acute lymphocytic leukemia) is a cancer of the blood and bone marrow. This type of cancer usually gets worse quickly if it is not treated.
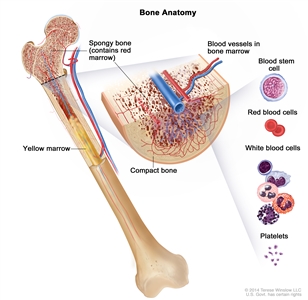
Anatomy of the bone. The bone is made up of compact bone, spongy bone, and bone marrow. Compact bone makes up the outer layer of the bone. Spongy bone is found mostly at the ends of bones and contains red marrow. Bone marrow is found in the center of most bones and has many blood vessels. There are two types of bone marrow: red and yellow. Red marrow contains blood stem cells that can become red blood cells, white blood cells, or platelets. Yellow marrow is made mostly of fat.
ALL is the most common type of cancer in children.
Leukemia may affect red blood cells, white blood cells, and platelets.
In a healthy child, the bone marrow makes blood stem cells (immature cells) that become mature blood cells over time. A blood stem cell may become a myeloid stem cell or a lymphoid stem cell.
A myeloid stem cell becomes one of three types of mature blood cells:
- Red blood cells that carry oxygen and other substances to all tissues of the body.
- Granulocytes, which are white blood cells that help fight infection and disease.
- Platelets that form blood clots to stop bleeding.
A lymphoid stem cell becomes a lymphoblast cell and then one of three types of lymphocytes (white blood cells):
- B lymphocytes that make antibodies to help fight infection.
- T lymphocytes that help B lymphocytes make the antibodies that help fight infection.
- Natural killer cells that attack cancer cells and viruses.
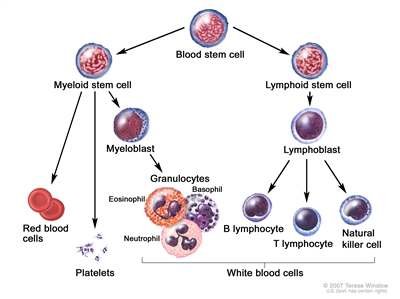
Blood cell development. A blood stem cell goes through several steps to become a red blood cell, platelet, or white blood cell.
In a child with ALL, too many stem cells become lymphoblasts, B lymphocytes, or T lymphocytes. These cells are also called leukemia cells. These leukemia cells do not work like normal lymphocytes and are not able to fight infection very well. Also, as the number of leukemia cells increases in the blood and bone marrow, there is less room for healthy white blood cells, red blood cells, and platelets. This may lead to infection, anemia, and easy bleeding.
This summary is about acute lymphoblastic leukemia in children, adolescents, and young adults. See the following PDQ summaries for information about other types of leukemia:
- Childhood Acute Myeloid Leukemia/Other Myeloid Malignancies Treatment
- Adult Acute Lymphoblastic Leukemia Treatment
- Chronic Lymphocytic Leukemia Treatment
- Acute Myeloid Leukemia Treatment
- Chronic Myelogenous Leukemia Treatment
- Hairy Cell Leukemia Treatment
Past treatment for cancer and certain genetic conditions affect the risk of having childhood ALL.
Anything that increases your risk of getting a disease is called a risk factor. Having a risk factor does not mean that you will get cancer; not having risk factors doesn't mean that you will not get cancer. Talk with your child's doctor if you think your child may be at risk.
Possible risk factors for ALL include the following:
- Being exposed to x-rays before birth.
- Being exposed to radiation.
- Past treatment with chemotherapy.
- Having certain genetic conditions, such as:
- Down syndrome.
- Neurofibromatosis type 1.
- Bloom syndrome.
- Fanconi anemia.
- Ataxia-telangiectasia.
- Li-Fraumeni syndrome.
- Constitutional mismatch repair deficiency (mutations in certain genes that stop DNA from repairing itself, which leads to the growth of cancers at an early age).
- Having certain changes in chromosomes or genes.
Signs of childhood ALL include fever and bruising.
These and other signs and symptoms may be caused by childhood ALL or by other conditions. Check with your child's doctor if your child has any of the following:
- Fever.
- Easy bruising or bleeding.
- Petechiae (flat, pinpoint, dark-red spots under the skin caused by bleeding).
- Weakness, feeling tired, or looking pale.
- Bone or joint pain.
- Shortness of breath.
- Painless lumps (swollen lymph nodes) in the neck, underarm, stomach, or groin.
- Pain or feeling of fullness below the ribs.
- Loss of appetite.
Tests that examine the blood and bone marrow are used to diagnose childhood ALL.
The following tests and procedures may be used to diagnose childhood ALL and find out if leukemia cells have spread to other parts of the body such as the brain or testicles:
- Physical exam and health history: An exam of the body to check general signs of health, including checking for signs of disease, such as lumps or anything else that seems unusual. A history of the patient's health habits and past illnesses and treatments will also be taken.
- Complete blood count (CBC) with differential: A procedure in which a sample of blood is drawn and checked for the following:
- The number of red blood cells and platelets.
- The number and type of white blood cells.
- The amount of hemoglobin (the protein that carries oxygen) in the red blood cells.
- The portion of the sample made up of red blood cells.
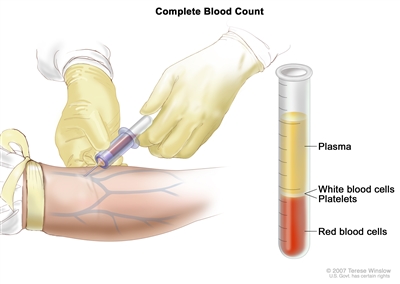
Complete blood count (CBC). Blood is collected by inserting a needle into a vein and allowing the blood to flow into a tube. The blood sample is sent to the laboratory and the red blood cells, white blood cells, and platelets are counted. The CBC is used to test for, diagnose, and monitor many different conditions. - Blood chemistry studies: A procedure in which a blood sample is checked to measure the amounts of certain substances released into the blood by organs and tissues in the body. An unusual (higher or lower than normal) amount of a substance can be a sign of disease.
- Bone marrow aspiration and biopsy: The removal of bone marrow and a small piece of bone by inserting a hollow needle into the hipbone or breastbone. A pathologist views the bone marrow and bone under a microscope to look for signs of cancer.
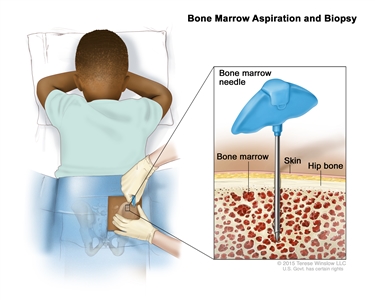
Bone marrow aspiration and biopsy. After a small area of skin is numbed, a bone marrow needle is inserted into the child's hip bone. Samples of blood, bone, and bone marrow are removed for examination under a microscope.The following tests are done on blood or the bone marrow tissue that is removed:
- Cytogenetic analysis: A laboratory test in which the chromosomes of cells in a sample of blood or bone marrow are counted and checked for any changes, such as broken, missing, rearranged, or extra chromosomes. Changes in certain chromosomes may be a sign of cancer. For example, in Philadelphia chromosome –positive ALL, part of one chromosome switches places with part of another chromosome. This is called the "Philadelphia chromosome." Cytogenetic analysis is used to help diagnose cancer, plan treatment, or find out how well treatment is working.
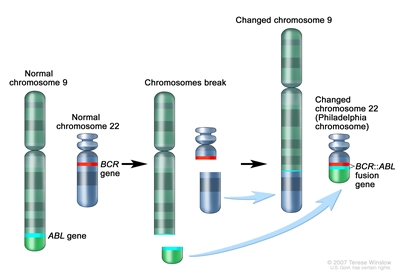
The Philadelphia (Ph) chromosome is an abnormal chromosome that is made when pieces of chromosomes 9 and 22 break off and trade places. The ABL gene from chromosome 9 joins to the BCR gene on chromosome 22 to form the BCR::ABL fusion gene. The changed chromosome 22 with the fusion gene on it is called the Ph chromosome. - Immunophenotyping: A laboratory test that uses antibodies to identify cancer cells based on the types of antigens or markers on the surface of the cells. This test is used to help diagnose specific types of leukemia. For example, the cancer cells are checked to see if they are B lymphocytes or T lymphocytes.
- Cytogenetic analysis: A laboratory test in which the chromosomes of cells in a sample of blood or bone marrow are counted and checked for any changes, such as broken, missing, rearranged, or extra chromosomes. Changes in certain chromosomes may be a sign of cancer. For example, in Philadelphia chromosome –positive ALL, part of one chromosome switches places with part of another chromosome. This is called the "Philadelphia chromosome." Cytogenetic analysis is used to help diagnose cancer, plan treatment, or find out how well treatment is working.
- Lumbar puncture: A procedure used to collect a sample of cerebrospinal fluid (CSF) from the spinal column. This is done by placing a needle between two bones in the spine and into the CSF around the spinal cord and removing a sample of the fluid. The sample of CSF is checked under a microscope for signs that leukemia cells have spread to the brain and spinal cord. This procedure is also called an LP or spinal tap.
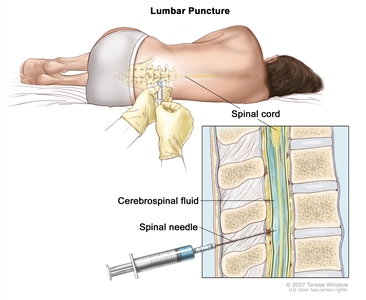
Lumbar puncture. A patient lies in a curled position on a table. After a small area on the lower back is numbed, a spinal needle (a long, thin needle) is inserted into the lower part of the spinal column to remove cerebrospinal fluid (CSF, shown in blue). The fluid may be sent to a laboratory for testing.This procedure is done after leukemia is diagnosed to find out if leukemia cells have spread to the brain and spinal cord. Intrathecal chemotherapy is given after the sample of fluid is removed to treat any leukemia cells that may have spread to the brain and spinal cord.
- Chest x-ray: An x-ray of the organs and bones inside the chest. An x-ray is a type of energy beam that can go through the body and onto film, making a picture of areas inside the body. The chest x-ray is done to see if leukemia cells have formed a mass in the middle of the chest.
Certain factors affect prognosis (chance of recovery) and treatment options.
The prognosis depends on:
- How quickly and how low the leukemia cell count drops after the first month of treatment.
- Age at the time of diagnosis, sex, race, and ethnic background.
- The number of white blood cells in the blood at the time of diagnosis.
- Whether the leukemia cells began from B lymphocytes or T lymphocytes.
- Whether there are certain changes in the chromosomes or genes of the leukemia cells.
- Whether the child has Down syndrome.
- Whether leukemia cells are found in the cerebrospinal fluid.
- The child's weight at the time of diagnosis and during treatment.
Treatment options depend on:
- Whether the leukemia cells began from B lymphocytes or T lymphocytes.
- Whether the child has standard-risk, high-risk, or very high–risk ALL.
- The age of the child at the time of diagnosis.
- Whether there are certain changes in the chromosomes of lymphocytes, such as the Philadelphia chromosome.
- Whether the child was treated with steroids before the start of induction therapy.
- How quickly and how low the leukemia cell count drops during treatment.
For leukemia that relapses (comes back) after treatment, the prognosis and treatment options depend partly on the following:
- How long it is between the time of diagnosis and when the leukemia comes back.
- Whether the leukemia comes back in the bone marrow or in other parts of the body.
Risk Groups for Childhood Acute Lymphoblastic Leukemia
In childhood ALL, risk groups are used to plan treatment.
There are three risk groups in childhood ALL. They are described as:
- Standard (low) risk: Includes children aged 1 to younger than 10 years who have a white blood cell count less than 50,000/µL at the time of diagnosis.
- High risk: Includes children 10 years and older and/or children who have a white blood cell count of 50,000/µL or more at the time of diagnosis.
- Very high risk: Includes children younger than age 1, children with certain changes in the genes, children who have a slow response to initial treatment, and children who have signs of leukemia after the first 4 weeks of treatment.
Other factors that affect the risk group include the following:
- Whether the leukemia cells began from B lymphocytes or T lymphocytes.
- Whether there are certain changes in the chromosomes or genes of the lymphocytes.
- How quickly and how low the leukemia cell count drops after initial treatment.
- Whether leukemia cells are found in the cerebrospinal fluid at the time of diagnosis.
It is important to know the risk group in order to plan treatment. Children with high-risk or very high–risk ALL usually receive more anticancer drugs and/or higher doses of anticancer drugs than children with standard-risk ALL.
Sometimes childhood ALL does not respond to treatment or comes back after treatment.
Refractory childhood ALL is cancer that does not respond to initial treatment.
Recurrent childhood ALL is cancer that has recurred (come back) after it has been treated. The leukemia may come back in the blood and bone marrow, brain, spinal cord, testicles, or other parts of the body.
Treatment Option Overview
There are different types of treatment for childhood acute lymphoblastic leukemia (ALL).
Different types of treatment are available for children with acute lymphoblastic leukemia (ALL). Some treatments are standard (the currently used treatment), and some are being tested in clinical trials. A treatment clinical trial is a research study meant to help improve current treatments or obtain information on new treatments for patients with cancer. When clinical trials show that a new treatment is better than the standard treatment, the new treatment may become the standard treatment.
Because cancer in children is rare, taking part in a clinical trial should be considered. Some clinical trials are open only to patients who have not started treatment.
Children with ALL should have their treatment planned by a team of doctors who are experts in treating childhood leukemia.
Treatment will be overseen by a pediatric oncologist, a doctor who specializes in treating children with cancer. The pediatric oncologist works with other pediatric health professionals who are experts in treating children with leukemia and who specialize in certain areas of medicine. These may include the following specialists:
- Pediatrician.
- Hematologist.
- Medical oncologist.
- Pediatric surgeon.
- Radiation oncologist.
- Pediatric intensivist.
- Neurologist.
- Neuroradiologist.
- Pediatric nurse specialist.
- Social worker.
- Rehabilitation specialist.
- Psychologist.
- Child-life specialist.
The treatment of childhood ALL usually has three phases.
The treatment of childhood ALL is done in phases:
- Remission induction: This is the first phase of treatment. The goal is to kill the leukemia cells in the blood and bone marrow. This puts the leukemia into remission.
- Consolidation /intensification: This is the second phase of treatment. It begins once the leukemia is in remission. The goal of consolidation/intensification therapy is to kill any leukemia cells that remain in the body and may cause a relapse.
- Maintenance: This is the third phase of treatment. The goal is to kill any remaining leukemia cells that may regrow and cause a relapse. Often the cancer treatments are given in lower doses than those used during the remission induction and consolidation/intensification phases. Not taking medication as ordered by the doctor during maintenance therapy increases the chance the cancer will come back. This is also called the continuation therapy phase.
Four types of standard treatment are used:
Chemotherapy
Chemotherapy is a cancer treatment that uses drugs to stop the growth of cancer cells, either by killing the cells or by stopping them from dividing. When chemotherapy is taken by mouth or injected into a vein or muscle, the drugs enter the bloodstream and can reach cancer cells throughout the body (systemic chemotherapy). When chemotherapy is placed directly into the cerebrospinal fluid (intrathecal), an organ, or a body cavity such as the abdomen, the drugs mainly affect cancer cells in those areas (regional chemotherapy). Combination chemotherapy is treatment using more than one anticancer drug.
The way the chemotherapy is given depends on the child's risk group. Children with high-risk ALL receive more anticancer drugs and higher doses of anticancer drugs than children with standard-risk ALL. Intrathecal chemotherapy is used to treat childhood ALL that has spread, or may spread, to the brain and spinal cord.
See
Radiation therapy
Radiation therapy is a cancer treatment that uses high-energy x-rays or other types of radiation to kill cancer cells or keep them from growing. External radiation therapy uses a machine outside the body to send radiation toward the area of the body with cancer.
External radiation therapy may be used to treat childhood ALL that has spread, or may spread, to the brain, spinal cord, or testicles. It may also be used to prepare the bone marrow for a stem cell transplant.
Chemotherapy with stem cell transplant
Chemotherapy is given to kill cancer cells. In children 3 years and older, total-body irradiation is given with chemotherapy. Healthy cells, including blood-forming cells, are also destroyed by the cancer treatment. Stem cell transplant is a treatment to replace the blood-forming cells. Stem cells (immature blood cells) are removed from the blood or bone marrow of a donor and are frozen and stored. After the patient completes chemotherapy and radiation therapy, the stored stem cells are thawed and given to the patient through an infusion. These stem cells grow into (and restore) the body's blood cells.
Stem cell transplant is rarely used as initial treatment for children and adolescents with ALL. It is used more often as part of treatment for ALL that relapses (comes back after treatment).
See
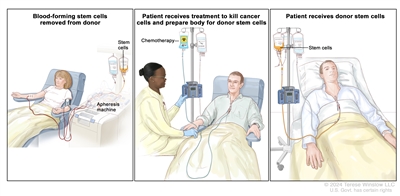
Stem cell transplant. (Step 1): Blood is taken from a vein in the arm of the donor. The blood flows through a machine that removes the stem cells. Then the blood is returned to the donor through a vein in the other arm. (Step 2): The patient receives chemotherapy to kill blood-forming cells. The patient may receive radiation therapy (not shown). (Step 3): The patient receives stem cells through a catheter placed into a blood vessel in the chest.
Targeted therapy
Targeted therapy is a type of treatment that uses drugs or other substances to identify and attack specific cancer cells. Targeted therapies usually cause less harm to normal cells than chemotherapy or radiation therapy do. There are different types of targeted therapy:
- Tyrosine kinase inhibitor (TKIs) therapy: This treatment blocks the enzyme, tyrosine kinase, that causes stem cells to develop into more white blood cells than the body needs. Imatinib mesylate and dasatinib are TKIs used in the treatment of children with Philadelphia chromosome –positive ALL. Ruxolitinib is a TKI that is being studied in the treatment of newly diagnosed high-risk ALL.
- Monoclonal antibodies: Monoclonal antibodies are immune system proteins made in the laboratory to treat many diseases, including cancer. As a cancer treatment, these antibodies can attach to a specific target on cancer cells or other cells that may help cancer cells grow. The antibodies are able to then kill the cancer cells, block their growth, or keep them from spreading. Monoclonal antibodies are given by infusion. They may be used alone or to carry drugs, toxins, or radioactive material directly to cancer cells. Blinatumomab and inotuzumab are monoclonal antibodies being studied in the treatment of refractory childhood ALL. Blinatumomab is also being studied in the treatment of standard-risk ALL. Nivolumab is a monoclonal antibody being studied in the treatment of relapsed childhood ALL.monoclonal antibodies: how monoclonal antibodies treat cancerHow do monoclonal antibodies work to treat cancer? This video shows how monoclonal antibodies, such as trastuzumab, pembrolizumab, and rituximab, block molecules cancer cells need to grow, flag cancer cells for destruction by the body's immune system, or deliver harmful substances to cancer cells.
- Proteasome inhibitor therapy: This treatment is a type of targeted therapy that blocks the action of proteasomes in cancer cells. Proteasomes remove proteins no longer needed by the cell. When the proteasomes are blocked, the proteins build up in the cell and may cause the cancer cell to die. Bortezomib is a type of proteasome inhibitor therapy used to treat relapsed childhood ALL. Bortezomib is also being studied in the treatment of standard-risk or high-risk patients.
New kinds of targeted therapies are also being studied in the treatment of childhood ALL.
See
Treatment is given to kill leukemia cells that have spread or may spread to the brain, spinal cord, or testicles.
Treatment to kill leukemia cells or prevent the spread of leukemia cells to the brain and spinal cord (central nervous system; CNS) is called CNS-directed therapy. Chemotherapy may be used to treat leukemia cells that have spread, or may spread, to the brain and spinal cord. Because standard doses of chemotherapy may not cross the blood-brain barrier to get into the fluid that surrounds the brain and spinal cord to reach the leukemia cells in the CNS, the cells are able to hide in the CNS. Systemic chemotherapy given in high doses or intrathecal chemotherapy (into the cerebrospinal fluid) is able to reach leukemia cells in the CNS. Sometimes external radiation therapy to the brain is also given.
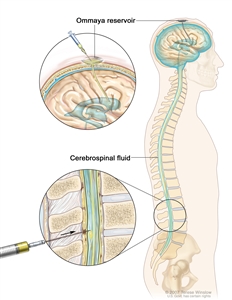
Intrathecal chemotherapy. Anticancer drugs are injected into the intrathecal space, which is the space that holds the cerebrospinal fluid (CSF, shown in blue). There are two different ways to do this. One way, shown in the top part of the figure, is to inject the drugs into an Ommaya reservoir (a dome-shaped container that is placed under the scalp during surgery; it holds the drugs as they flow through a small tube into the brain). The other way, shown in the bottom part of the figure, is to inject the drugs directly into the CSF in the lower part of the spinal column, after a small area on the lower back is numbed.
These treatments are given in addition to treatment that is used to kill leukemia cells in the rest of the body. All children with ALL receive CNS-directed therapy as part of induction therapy and consolidation/intensification therapy and sometimes during maintenance therapy.
If the leukemia cells spread to the testicles, treatment includes high doses of systemic chemotherapy and sometimes radiation therapy.
New types of treatment are being tested in clinical trials.
This summary section describes treatments that are being studied in clinical trials. It may not mention every new treatment being studied. Information about clinical trials is available from the
Immunotherapy
Immunotherapy is a treatment that uses the patient's immune system to fight cancer. Substances made by the body or made in a laboratory are used to boost, direct, or restore the body's natural defenses against cancer. This cancer treatment is a type of biologic therapy.
- CAR T-cell therapy: This treatment changes the patient's T cells (a type of immune system cell) so they will attack certain proteins on the surface of cancer cells. T cells are taken from the patient and special receptors are added to their surface in the laboratory. The changed cells are called chimeric antigen receptor (CAR) T cells. The CAR T cells are grown in the laboratory and given to the patient by infusion. The CAR T cells multiply in the patient's blood and attack cancer cells. CAR T-cell therapy is being studied in the treatment of childhood ALL that has relapsed (come back) a second time.
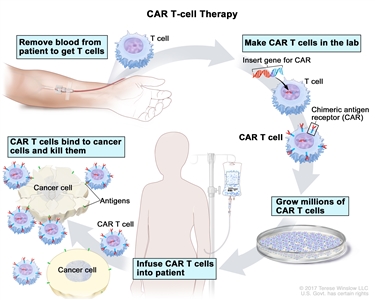
CAR T-cell therapy. A type of treatment in which a patient's T cells (a type of immune cell) are changed in the laboratory so they will bind to cancer cells and kill them. Blood from a vein in the patient's arm flows through a tube to an apheresis machine (not shown), which removes the white blood cells, including the T cells, and sends the rest of the blood back to the patient. Then, the gene for a special receptor called a chimeric antigen receptor (CAR) is inserted into the T cells in the laboratory. Millions of the CAR T cells are grown in the laboratory and then given to the patient by infusion. The CAR T cells are able to bind to an antigen on the cancer cells and kill them.
Treatment for childhood acute lymphoblastic leukemia may cause side effects.
To learn more about side effects that begin during treatment for cancer, visit
Regular follow-up exams are very important. Side effects from cancer treatment that begin after treatment and continue for months or years are called late effects.
Late effects of cancer treatment may include the following:
- Physical problems, including problems with the heart, blood vessels, liver, or bones, and fertility. When dexrazoxane is given with chemotherapy drugs called anthracyclines, the risk of late heart effects is lessened.
- Changes in mood, feelings, thinking, learning, or memory. Children younger than 4 years who have received radiation therapy to the brain have a higher risk of these effects.
- Second cancers (new types of cancer) or other conditions, such as brain tumors, thyroid cancer, acute myeloid leukemia, and myelodysplastic syndrome.
Some late effects may be treated or controlled. It is important to talk with your child's doctors about the possible late effects caused by some treatments. See the PDQ summary on Late Effects of Treatment for Childhood Cancer.
Patients may want to think about taking part in a clinical trial.
For some patients, taking part in a clinical trial may be the best treatment choice. Clinical trials are part of the cancer research process. Clinical trials are done to find out if new cancer treatments are safe and effective or better than the standard treatment.
Many of today's standard treatments for cancer are based on earlier clinical trials. Patients who take part in a clinical trial may receive the standard treatment or be among the first to receive a new treatment.
Patients who take part in clinical trials also help improve the way cancer will be treated in the future. Even when clinical trials do not lead to effective new treatments, they often answer important questions and help move research forward.
Patients can enter clinical trials before, during, or after starting their cancer treatment.
Some clinical trials only include patients who have not yet received treatment. Other trials test treatments for patients whose cancer has not gotten better. There are also clinical trials that test new ways to stop cancer from recurring (coming back) or reduce the side effects of cancer treatment.
Clinical trials are taking place in many parts of the country. Information about clinical trials supported by NCI can be found on NCI's
Follow-up tests may be needed.
As your child goes through treatment, they will have follow-up tests or check-ups. Some tests that were done to diagnose or stage the cancer may be repeated to see how well the treatment is working. Decisions about whether to continue, change, or stop treatment may be based on the results of these tests.
Some of the tests will continue to be done from time to time after treatment has ended. The results of these tests can show if your child's condition has changed or if the cancer has recurred (come back).
Bone marrow aspiration and biopsy is done during all phases of treatment to see how well the treatment is working.
Treatment of Childhood Acute Lymphoblastic Leukemia (Standard Risk)
For information about the treatments listed below, see the Treatment Option Overview section.
The treatment of newly diagnosed standard-risk childhood acute lymphoblastic leukemia (ALL) during the remission induction, consolidation /intensification, and maintenance phases always includes combination chemotherapy. When children with a poor response to treatment are in remission after remission induction therapy, a stem cell transplant using stem cells from a donor may be done. When children with a poor response to treatment are not in remission after remission induction therapy, further treatment is usually the same treatment given to children with high-risk ALL.
Intrathecal chemotherapy is given to prevent the spread of leukemia cells to the brain and spinal cord.
Treatments being studied in clinical trials for standard-risk ALL include combination chemotherapy with or without targeted therapy with a monoclonal antibody (blinatumomab).
Use our
Treatment of Childhood Acute Lymphoblastic Leukemia (High Risk)
For information about the treatments listed below, see the Treatment Option Overview section.
The treatment of newly diagnosed high-risk childhood acute lymphoblastic leukemia (ALL) during the remission induction, consolidation /intensification, and maintenance phases always includes combination chemotherapy. Children in the high-risk ALL group are given more anticancer drugs and higher doses of anticancer drugs, especially during the consolidation/intensification phase, than children in the standard-risk group.
Intrathecal and systemic chemotherapy are given to prevent or treat the spread of leukemia cells to the brain and spinal cord. Sometimes radiation therapy to the brain is also given.
Treatments being studied in clinical trials for high-risk ALL include new chemotherapy regimens with or without targeted therapy or stem cell transplant. Chimeric antigen receptor (CAR) T-cell therapy is also being studied.
Use our
Treatment of Childhood Acute Lymphoblastic Leukemia (Very High Risk)
For information about the treatments listed below, see the Treatment Option Overview section.
The treatment of newly diagnosed very high–risk childhood acute lymphoblastic leukemia (ALL) during the remission induction, consolidation /intensification, and maintenance phases always includes combination chemotherapy. Children in the very high–risk ALL group are given more anticancer drugs than children in the high-risk group. It is not clear whether a stem cell transplant during first remission will help the child live longer.
Intrathecal and systemic chemotherapy are given to prevent or treat the spread of leukemia cells to the brain and spinal cord. Sometimes radiation therapy to the brain is also given.
Treatments being studied in clinical trials for very high–risk ALL include new chemotherapy regimens with or without targeted therapy.
Use our
Treatment of Childhood Acute Lymphoblastic Leukemia (Special Groups)
For information about the treatments listed below, see the Treatment Option Overview section.
T-cell childhood acute lymphoblastic leukemia
The treatment of newly diagnosed T-cell childhood acute lymphoblastic leukemia (T-ALL) during the remission induction, consolidation /intensification, and maintenance phases always includes combination chemotherapy. Children with T-ALL are given more anticancer drugs and higher doses of anticancer drugs than children in the newly diagnosed standard-risk group.
Intrathecal and systemic chemotherapy are given to prevent or treat the spread of leukemia cells to the brain and spinal cord. Sometimes radiation therapy to the brain is also given.
Infants with ALL
The treatment of infants with newly diagnosed ALL during the remission induction, consolidation /intensification, and maintenance phases always includes combination chemotherapy. Infants with ALL are given different anticancer drugs and higher doses of anticancer drugs than children 1 year and older in the standard-risk group. It is not clear whether a stem cell transplant during first remission will help the child live longer.
Intrathecal and systemic chemotherapy are given to prevent or treat the spread of leukemia cells to the brain and spinal cord.
Children 10 years and older and adolescents with ALL
The treatment of newly diagnosed ALL in children and adolescents (10 years and older) during the remission induction, consolidation /intensification, and maintenance phases always includes combination chemotherapy. Children 10 years and older and adolescents with ALL are given more anticancer drugs and higher doses of anticancer drugs than children in the standard-risk group.
Intrathecal and systemic chemotherapy are given to prevent or treat the spread of leukemia cells to the brain and spinal cord. Sometimes radiation therapy to the brain is also given.
Treatments being studied in clinical trials for children 10 years and older and adolescents with ALL include new anticancer agents and chemotherapy regimens with or without targeted therapy. Chimeric antigen receptor (CAR) T-cell therapy is also being studied.
Philadelphia chromosome–positive ALL
The treatment of newly diagnosed Philadelphia chromosome –positive childhood ALL during the remission induction, consolidation /intensification, and maintenance phases may include the following:
- Combination chemotherapy and targeted therapy with a tyrosine kinase inhibitor (imatinib mesylate or dasatinib) with or without a stem cell transplant using stem cells from a donor.
Intrathecal and systemic chemotherapy are given to prevent or treat the spread of leukemia cells to the brain and spinal cord.
Treatments being studied in clinical trials for Philadelphia chromosome–positive childhood ALL include a new regimen of targeted therapy (imatinib mesylate) and combination chemotherapy with or without a stem cell transplant.
Use our
Treatment of Refractory or Relapsed Childhood Acute Lymphoblastic Leukemia
For information about the treatments listed below, see the Treatment Option Overview section.
Standard treatment of relapsed childhood acute lymphoblastic leukemia (ALL) that comes back in the bone marrow may include the following:
- Combination chemotherapy with or without targeted therapy (bortezomib).
- Stem cell transplant, using stem cells from a donor.
There is no standard treatment for the treatment of refractory childhood ALL.
Other treatments for refractory or relapsed childhood ALL may include the following:
- Targeted therapy (blinatumomab or inotuzumab).
- Chimeric antigen receptor (CAR) T-cell therapy.
Standard treatment of relapsed childhood ALL that comes back outside the bone marrow may include the following:
- Systemic chemotherapy and intrathecal chemotherapy with radiation therapy to the brain and/or spinal cord for cancer that comes back in the brain and spinal cord only.
- Stem cell transplant for cancer that has recurred in the brain and/or spinal cord.
- Combination chemotherapy and radiation therapy for cancer that comes back in the testicles only.
Some of the treatments being studied in clinical trials for relapsed childhood ALL include:
- Targeted therapy (blinatumomab) alone or combined with another targeted therapy (nivolumab).
- A new type of chemotherapy drug.
- CAR T-cell therapy.
To Learn More About Childhood Acute Lymphoblastic Leukemia
For more information from the National Cancer Institute about childhood acute lymphoblastic leukemia, see the following:
-
Computed Tomography (CT) Scans and Cancer -
Drugs Approved for Acute Lymphoblastic Leukemia -
Blood-Forming Stem Cell Transplants -
Targeted Cancer Therapies -
CAR T Cells: Engineering Patients' Immune Cells to Treat Their Cancers
For more childhood cancer information and other general cancer resources, visit:
-
About Cancer -
Childhood Cancers -
CureSearch for Children's Cancer - Late Effects of Treatment for Childhood Cancer
-
Adolescents and Young Adults with Cancer -
Children with Cancer: A Guide for Parents -
Cancer in Children and Adolescents -
Staging -
Coping with Cancer -
Questions to Ask Your Doctor about Cancer -
For Survivors and Caregivers
About This PDQ Summary
About PDQ
Physician Data Query (PDQ) is the National Cancer Institute's (NCI's) comprehensive cancer information database. The PDQ database contains summaries of the latest published information on cancer prevention, detection, genetics, treatment, supportive care, and complementary and alternative medicine. Most summaries come in two versions. The health professional versions have detailed information written in technical language. The patient versions are written in easy-to-understand, nontechnical language. Both versions have cancer information that is accurate and up to date and most versions are also available in
PDQ is a service of the NCI. The NCI is part of the National Institutes of Health (NIH). NIH is the federal government's center of biomedical research. The PDQ summaries are based on an independent review of the medical literature. They are not policy statements of the NCI or the NIH.
Purpose of This Summary
This PDQ cancer information summary has current information about the treatment of childhood acute lymphoblastic leukemia. It is meant to inform and help patients, families, and caregivers. It does not give formal guidelines or recommendations for making decisions about health care.
Reviewers and Updates
Editorial Boards write the PDQ cancer information summaries and keep them up to date. These Boards are made up of experts in cancer treatment and other specialties related to cancer. The summaries are reviewed regularly and changes are made when there is new information. The date on each summary ("Updated") is the date of the most recent change.
The information in this patient summary was taken from the health professional version, which is reviewed regularly and updated as needed, by the
Clinical Trial Information
A clinical trial is a study to answer a scientific question, such as whether one treatment is better than another. Trials are based on past studies and what has been learned in the laboratory. Each trial answers certain scientific questions in order to find new and better ways to help cancer patients. During treatment clinical trials, information is collected about the effects of a new treatment and how well it works. If a clinical trial shows that a new treatment is better than one currently being used, the new treatment may become "standard." Patients may want to think about taking part in a clinical trial. Some clinical trials are open only to patients who have not started treatment.
Clinical trials can be found online at
Permission to Use This Summary
PDQ is a registered trademark. The content of PDQ documents can be used freely as text. It cannot be identified as an NCI PDQ cancer information summary unless the whole summary is shown and it is updated regularly. However, a user would be allowed to write a sentence such as "NCI's PDQ cancer information summary about breast cancer prevention states the risks in the following way: [include excerpt from the summary]."
The best way to cite this PDQ summary is:
PDQ® Pediatric Treatment Editorial Board. PDQ Childhood Acute Lymphoblastic Leukemia Treatment. Bethesda, MD: National Cancer Institute. Updated <MM/DD/YYYY>. Available at:
Images in this summary are used with permission of the author(s), artist, and/or publisher for use in the PDQ summaries only. If you want to use an image from a PDQ summary and you are not using the whole summary, you must get permission from the owner. It cannot be given by the National Cancer Institute. Information about using the images in this summary, along with many other images related to cancer can be found in
Disclaimer
The information in these summaries should not be used to make decisions about insurance reimbursement. More information on insurance coverage is available on Cancer.gov on the
Contact Us
More information about contacting us or receiving help with the Cancer.gov website can be found on our
Last Revised: 2022-09-02
If you want to know more about cancer and how it is treated, or if you wish to know about clinical trials for your type of cancer, you can call the NCI's Cancer Information Service at 1-800-422-6237, toll free. A trained information specialist can talk with you and answer your questions.
This information does not replace the advice of a doctor. Ignite Healthwise, LLC, disclaims any warranty or liability for your use of this information. Your use of this information means that you agree to the
Healthwise, Healthwise for every health decision, and the Healthwise logo are trademarks of Ignite Healthwise, LLC.
Page Footer
I want to...
Audiences
Secure Member Sites
The Cigna Group Information
Disclaimer
Individual and family medical and dental insurance plans are insured by Cigna Health and Life Insurance Company (CHLIC), Cigna HealthCare of Arizona, Inc., Cigna HealthCare of Illinois, Inc., Cigna HealthCare of Georgia, Inc., Cigna HealthCare of North Carolina, Inc., Cigna HealthCare of South Carolina, Inc., and Cigna HealthCare of Texas, Inc. Group health insurance and health benefit plans are insured or administered by CHLIC, Connecticut General Life Insurance Company (CGLIC), or their affiliates (see
All insurance policies and group benefit plans contain exclusions and limitations. For availability, costs and complete details of coverage, contact a licensed agent or Cigna sales representative. This website is not intended for residents of New Mexico.