Shop for Plans
Shop for your own coverage
Plans through your employer
Learn about the medical, dental, pharmacy, behavioral, and voluntary benefits your employer may offer.
Learn
Living or working abroad?
Hereditary Diffuse Gastric Cancer (PDQ®): Genetics - Health Professional Information [NCI]
Introduction
Hereditary diffuse gastric cancer (HDGC) is an autosomal dominant hereditary cancer syndrome that increases an individual's risk to develop diffuse gastric cancer and lobular breast cancer. HDGC is defined by the presence of germline pathogenic variants in the CDH1 gene, which codes for the cell–cell adhesion junction protein, E-cadherin. In patients with CDH1 pathogenic variants, early stage gastric cancer is characterized by intramucosal foci of signet ring cell carcinoma while advanced gastric cancer presents as poorly cohesive diffuse type carcinoma with very few typical signet ring cells.[
Pathogenic variants in a second gene, CTNNA1, are found in a small number of families with HDGC. This gene codes for an adhesion junction protein, catenin alpha-1.[
References:
- van der Post RS, Gullo I, Oliveira C, et al.: Histopathological, Molecular, and Genetic Profile of Hereditary Diffuse Gastric Cancer: Current Knowledge and Challenges for the Future. Adv Exp Med Biol 908: 371-91, 2016.
- Pharoah PD, Guilford P, Caldas C, et al.: Incidence of gastric cancer and breast cancer in CDH1 (E-cadherin) mutation carriers from hereditary diffuse gastric cancer families. Gastroenterology 121 (6): 1348-53, 2001.
- Roberts ME, Ranola JMO, Marshall ML, et al.: Comparison of CDH1 Penetrance Estimates in Clinically Ascertained Families vs Families Ascertained for Multiple Gastric Cancers. JAMA Oncol 5 (9): 1325-1331, 2019.
- Xicola RM, Li S, Rodriguez N, et al.: Clinical features and cancer risk in families with pathogenic CDH1 variants irrespective of clinical criteria. J Med Genet 56 (12): 838-843, 2019.
- Hansford S, Kaurah P, Li-Chang H, et al.: Hereditary Diffuse Gastric Cancer Syndrome: CDH1 Mutations and Beyond. JAMA Oncol 1 (1): 23-32, 2015.
- Delgado E, León-Ponte M, Villahermosa ML, et al.: Analysis of HIV type 1 protease and reverse transcriptase sequences from Venezuela for drug resistance-associated mutations and subtype classification: a UNAIDS study. AIDS Res Hum Retroviruses 17 (8): 753-8, 2001.
- Caldas C, Carneiro F, Lynch HT, et al.: Familial gastric cancer: overview and guidelines for management. J Med Genet 36 (12): 873-80, 1999.
- Corso G, Intra M, Trentin C, et al.: CDH1 germline mutations and hereditary lobular breast cancer. Fam Cancer 15 (2): 215-9, 2016.
- van der Post RS, Vogelaar IP, Carneiro F, et al.: Hereditary diffuse gastric cancer: updated clinical guidelines with an emphasis on germline CDH1 mutation carriers. J Med Genet 52 (6): 361-74, 2015.
- Majewski IJ, Kluijt I, Cats A, et al.: An α-E-catenin (CTNNA1) mutation in hereditary diffuse gastric cancer. J Pathol 229 (4): 621-9, 2013.
- Benusiglio PR, Colas C, Guillerm E, et al.: Clinical implications of CTNNA1 germline mutations in asymptomatic carriers. Gastric Cancer 22 (4): 899-903, 2019.
- Coudert M, Drouet Y, Delhomelle H, et al.: First estimates of diffuse gastric cancer risks for carriers of CTNNA1 germline pathogenic variants. J Med Genet 59 (12): 1189-1195, 2022.
- Clark DF, Michalski ST, Tondon R, et al.: Loss-of-function variants in CTNNA1 detected on multigene panel testing in individuals with gastric or breast cancer. Genet Med 22 (5): 840-846, 2020.
Genetic Testing Criteria for Hereditary Diffuse Gastric Cancer (HDGC)
The International Gastric Cancer Linkage Consortium's (IGCLC) 2020 HDGC genetic testing guidelines are summarized below.[
When an individual meets HDGC genetic testing criteria but genetic testing does not reveal a CDH1pathogenic variant, CTNNA1 genetic testing is recommended.
Personal History Criteria
- Diffuse gastric cancer diagnosed at age 49 years or younger.
- Diffuse gastric cancer diagnosed at any age in individuals of Māori descent.
- Diffuse gastric cancer and cleft lip or cleft palate diagnosed in the same individual.
- Diffuse gastric cancer and a first-degree relative (FDR) with cleft lip or cleft palate.
- Diffuse gastric cancer and lobular breast cancer, both diagnosed in the same individual at age 69 years or younger.
- Bilateral lobular breast cancer diagnosed at age 69 years or younger.
- Gastric signet ring cell carcinoma in situ or pagetoid spread of gastric signet ring cells diagnosed at age 49 years or younger.
Family History Criteria a
- 2 or more cases of gastric cancer (with at least 1 diffuse gastric cancer case) occurring in the same family, regardless of age.
- 1 or more cases of diffuse gastric cancer diagnosed at any age, and 1 or more cases of lobular breast cancer diagnosed at age 69 years or younger (these cancers must occur in different family members).
- 2 or more cases of lobular breast cancer diagnosed in family members at age 49 years or younger.
a Family members must be FDRs or second-degree relatives of each other. The IGCLC recommends that affected family members undergo genetic testing, when possible. Providers may want to consider testing tissue (tumor tissue or healthy tissue) from deceased, affected relatives if a family does not have living individuals with breast or gastric cancers. If none of these options are feasible, providers can consider performing genetic testing in unaffected family members. Diffuse gastric cancer is generally diagnosed at an advanced stage (i.e., stage III or stage IV).[
References:
- Blair VR, McLeod M, Carneiro F, et al.: Hereditary diffuse gastric cancer: updated clinical practice guidelines. Lancet Oncol 21 (8): e386-e397, 2020.
Definition and Management of Hereditary Diffuse Gastric Cancer (HDGC)–Like Families
CDH1germline pathogenic variants are found in 30% to 40% of families with clinically defined HDGC from different ethnic backgrounds.[
In HDGC-like families, affected individuals and their FDRs may be considered for yearly endoscopic surveillance for at least 2 years after receiving a negative CDH1 genetic test result.[
References:
- Lynch HT, Grady W, Suriano G, et al.: Gastric cancer: new genetic developments. J Surg Oncol 90 (3): 114-33; discussion 133, 2005.
- Suriano G, Oliveira C, Ferreira P, et al.: Identification of CDH1 germline missense mutations associated with functional inactivation of the E-cadherin protein in young gastric cancer probands. Hum Mol Genet 12 (5): 575-82, 2003.
- Blair VR, McLeod M, Carneiro F, et al.: Hereditary diffuse gastric cancer: updated clinical practice guidelines. Lancet Oncol 21 (8): e386-e397, 2020.
Genetics
Hereditary diffuse gastric cancer (HDGC) was first identified in a large, indigenous Māori kindred from New Zealand. These individuals had a germline pathogenic variant in the CDH1gene. [
Molecular Biology
In 1995, the CDH1 (cadherin-1) gene was mapped to chromosome 16q22.1.[
In 1998, it was determined that germline, heterozygous pathogenic variants in the CDH1 tumor suppressor gene can cause HDGC.[
A systematic review of the literature from 1998 to 2021 identified 571 germline CDH1 pathogenic variants, with 387 (67.8%) variants reported in 108 different families.[
Genotype-Phenotype Correlations
CDH1 pathogenic variants demonstrate considerable heterogeneity with the types of cancers and the ages of cancer onset seen in carriers. However, a review of available CDH1 literature from 1985 to 2018 did not definitively establish genotype -phenotype correlations that could help direct patient management. This review found that CDH1 pathogenic variants are evenly distributed throughout the gene without a preferential variant type (i.e., nonsense, missense, etc.) or location.[
Possible CDH1 genotype-phenotype correlations have been observed in families with HDGC and cleft lip or cleft palate. However, it is unclear which CDH1 pathogenic variants lead to cleft lip or cleft palate. In 2006, CDH1 splice site variants were found in two families with HDGC and cleft lip or cleft palate. The splice site variants resulted in complex, aberrant splicing in lymphocytes.[
The term, hereditary lobular breast cancer, was created by the International Gastric Cancer Linkage Consortium to describe individuals with a CDH1 pathogenic variant and personal/family histories of lobular breast cancer, but no known personal/family histories of gastric cancer.[
Overall, there are insufficient data regarding possible CDH1 genotype-phenotype correlations. These findings are not substantial enough to guide individualized CDH1 cancer risk assessment and management.
References:
- Guilford P, Hopkins J, Harraway J, et al.: E-cadherin germline mutations in familial gastric cancer. Nature 392 (6674): 402-5, 1998.
- Hansford S, Kaurah P, Li-Chang H, et al.: Hereditary Diffuse Gastric Cancer Syndrome: CDH1 Mutations and Beyond. JAMA Oncol 1 (1): 23-32, 2015.
- Brooks-Wilson AR, Kaurah P, Suriano G, et al.: Germline E-cadherin mutations in hereditary diffuse gastric cancer: assessment of 42 new families and review of genetic screening criteria. J Med Genet 41 (7): 508-17, 2004.
- Kaurah P, MacMillan A, Boyd N, et al.: Founder and recurrent CDH1 mutations in families with hereditary diffuse gastric cancer. JAMA 297 (21): 2360-72, 2007.
- Berx G, Staes K, van Hengel J, et al.: Cloning and characterization of the human invasion suppressor gene E-cadherin (CDH1). Genomics 26 (2): 281-9, 1995.
- Yelskaya Z, Bacares R, Salo-Mullen E, et al.: CDH1 Missense Variant c.1679C>G (p.T560R) Completely Disrupts Normal Splicing through Creation of a Novel 5' Splice Site. PLoS One 11 (11): e0165654, 2016.
- Paredes J, Figueiredo J, Albergaria A, et al.: Epithelial E- and P-cadherins: role and clinical significance in cancer. Biochim Biophys Acta 1826 (2): 297-311, 2012.
- van Roy F, Berx G: The cell-cell adhesion molecule E-cadherin. Cell Mol Life Sci 65 (23): 3756-88, 2008.
- Shapiro L, Fannon AM, Kwong PD, et al.: Structural basis of cell-cell adhesion by cadherins. Nature 374 (6520): 327-37, 1995.
- Aberle H, Schwartz H, Kemler R: Cadherin-catenin complex: protein interactions and their implications for cadherin function. J Cell Biochem 61 (4): 514-23, 1996.
- Okamoto R, Irie K, Yamada A, et al.: Recruitment of E-cadherin associated with alpha- and beta-catenins and p120ctn to the nectin-based cell-cell adhesion sites by the action of 12-O-tetradecanoylphorbol-13-acetate in MDCK cells. Genes Cells 10 (5): 435-45, 2005.
- Kim NG, Koh E, Chen X, et al.: E-cadherin mediates contact inhibition of proliferation through Hippo signaling-pathway components. Proc Natl Acad Sci U S A 108 (29): 11930-5, 2011.
- Figueiredo J, Melo S, Carneiro P, et al.: Clinical spectrum and pleiotropic nature of CDH1 germline mutations. J Med Genet 56 (4): 199-208, 2019.
- Karam R, Carvalho J, Bruno I, et al.: The NMD mRNA surveillance pathway downregulates aberrant E-cadherin transcripts in gastric cancer cells and in CDH1 mutation carriers. Oncogene 27 (30): 4255-60, 2008.
- Luo X, Maciaszek JL, Thompson BA, et al.: Optimising clinical care through CDH1-specific germline variant curation: improvement of clinical assertions and updated curation guidelines. J Med Genet 60 (6): 568-575, 2023.
- Richards S, Aziz N, Bale S, et al.: Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 17 (5): 405-24, 2015.
- Garcia-Pelaez J, Barbosa-Matos R, Lobo S, et al.: Genotype-first approach to identify associations between CDH1 germline variants and cancer phenotypes: a multicentre study by the European Reference Network on Genetic Tumour Risk Syndromes. Lancet Oncol 24 (1): 91-106, 2023.
- Corso G, Tagliaferri V, Massari G, et al.: CDH1 mutations recurrence and global clustering in genetically tested families with hereditary diffuse gastric cancer syndrome: results from a systematic study. Fam Cancer 22 (2): 187-192, 2023.
- Pan Z, Fu Z, Luo C, et al.: CDH1 germline mutations in a Chinese cohort with hereditary diffuse gastric cancer. J Cancer Res Clin Oncol 148 (8): 2145-2151, 2022.
- Lo W, Zhu B, Sabesan A, et al.: Associations of CDH1 germline variant location and cancer phenotype in families with hereditary diffuse gastric cancer (HDGC). J Med Genet 56 (6): 370-379, 2019.
- Corso G, Magnoni F, Massari G, et al.: CDH1 germline mutations in healthy individuals from families with the hereditary diffuse gastric cancer syndrome. J Med Genet 59 (4): 313-317, 2022.
- Frebourg T, Oliveira C, Hochain P, et al.: Cleft lip/palate and CDH1/E-cadherin mutations in families with hereditary diffuse gastric cancer. J Med Genet 43 (2): 138-42, 2006.
- Corso G, Marrelli D, Pascale V, et al.: Frequency of CDH1 germline mutations in gastric carcinoma coming from high- and low-risk areas: metanalysis and systematic review of the literature. BMC Cancer 12: 8, 2012.
- Obermair F, Rammer M, Burghofer J, et al.: Cleft lip/palate and hereditary diffuse gastric cancer: report of a family harboring a CDH1 c.687 + 1G > A germline mutation and review of the literature. Fam Cancer 18 (2): 253-260, 2019.
- Selvanathan A, Nixon CY, Zhu Y, et al.: CDH1 Mutation Distribution and Type Suggests Genetic Differences between the Etiology of Orofacial Clefting and Gastric Cancer. Genes (Basel) 11 (4): , 2020.
- Blair VR, McLeod M, Carneiro F, et al.: Hereditary diffuse gastric cancer: updated clinical practice guidelines. Lancet Oncol 21 (8): e386-e397, 2020.
- Gamble LA, Rossi A, Fasaye GA, et al.: Association Between Hereditary Lobular Breast Cancer Due to CDH1 Variants and Gastric Cancer Risk. JAMA Surg 157 (1): 18-22, 2022.
Clinical Manifestations
Gastric Cancer Risk
In CDH1pathogenic variant carriers, lifetime risk of diffuse gastric cancer by age 80 years ranges from 37% to 70% in men and 25% to 83% in women.[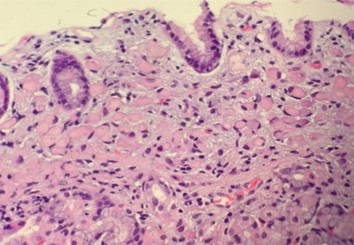
Figure 1. High-power image of a gastric biopsy tissue sample showing signet ring cell carcinoma. Note the absence of gland formation, which indicates that this lesion is poorly-differentiated carcinoma. Also note the overlying epithelium, which although effaced by the presence of underlying tumor, is otherwise normal.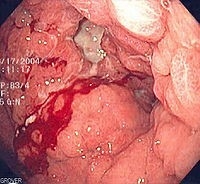
Figure 2. Endoscopic image of diffuse gastric cancer with typical linitis plastica involvement. This occurs at an advanced stage. The gastric mucosa and submucosa are diffusely infiltrated such that the entire stomach becomes stiff, losing contractility and the ability to digest food. This leads to the typical symptoms of nausea and vomiting. Biopsy is generally informative, but a very superficial biopsy may be negative for cancer cells. Hence, it is imperative for pathologists to carefully search biopsy specimens for isolated signet ring cells.
Breast Cancer Risk
In the general population, most breast cancers are ductal, while about 10% are lobular.[
References:
- Pharoah PD, Guilford P, Caldas C, et al.: Incidence of gastric cancer and breast cancer in CDH1 (E-cadherin) mutation carriers from hereditary diffuse gastric cancer families. Gastroenterology 121 (6): 1348-53, 2001.
- Roberts ME, Ranola JMO, Marshall ML, et al.: Comparison of CDH1 Penetrance Estimates in Clinically Ascertained Families vs Families Ascertained for Multiple Gastric Cancers. JAMA Oncol 5 (9): 1325-1331, 2019.
- Xicola RM, Li S, Rodriguez N, et al.: Clinical features and cancer risk in families with pathogenic CDH1 variants irrespective of clinical criteria. J Med Genet 56 (12): 838-843, 2019.
- Hansford S, Kaurah P, Li-Chang H, et al.: Hereditary Diffuse Gastric Cancer Syndrome: CDH1 Mutations and Beyond. JAMA Oncol 1 (1): 23-32, 2015.
- Delgado E, León-Ponte M, Villahermosa ML, et al.: Analysis of HIV type 1 protease and reverse transcriptase sequences from Venezuela for drug resistance-associated mutations and subtype classification: a UNAIDS study. AIDS Res Hum Retroviruses 17 (8): 753-8, 2001.
- Caldas C, Carneiro F, Lynch HT, et al.: Familial gastric cancer: overview and guidelines for management. J Med Genet 36 (12): 873-80, 1999.
- Li CI, Anderson BO, Daling JR, et al.: Trends in incidence rates of invasive lobular and ductal breast carcinoma. JAMA 289 (11): 1421-4, 2003.
- Corso G, Intra M, Trentin C, et al.: CDH1 germline mutations and hereditary lobular breast cancer. Fam Cancer 15 (2): 215-9, 2016.
- Corso G, Figueiredo J, La Vecchia C, et al.: Hereditary lobular breast cancer with an emphasis on E-cadherin genetic defect. J Med Genet 55 (7): 431-441, 2018.
- Masciari S, Larsson N, Senz J, et al.: Germline E-cadherin mutations in familial lobular breast cancer. J Med Genet 44 (11): 726-31, 2007.
- Kaurah P, MacMillan A, Boyd N, et al.: Founder and recurrent CDH1 mutations in families with hereditary diffuse gastric cancer. JAMA 297 (21): 2360-72, 2007.
- Mouabbi JA, Hassan A, Lim B, et al.: Invasive lobular carcinoma: an understudied emergent subtype of breast cancer. Breast Cancer Res Treat 193 (2): 253-264, 2022.
- Pilonis ND, Tischkowitz M, Fitzgerald RC, et al.: Hereditary Diffuse Gastric Cancer: Approaches to Screening, Surveillance, and Treatment. Annu Rev Med 72: 263-280, 2021.
- Rakha EA, El-Sayed ME, Powe DG, et al.: Invasive lobular carcinoma of the breast: response to hormonal therapy and outcomes. Eur J Cancer 44 (1): 73-83, 2008.
- Lamovec J, Bracko M: Metastatic pattern of infiltrating lobular carcinoma of the breast: an autopsy study. J Surg Oncol 48 (1): 28-33, 1991.
- National Comprehensive Cancer Network: NCCN Clinical Practice Guidelines in Oncology: Breast Cancer. Version 4.2023. Plymouth Meeting, Pa: National Comprehensive Cancer Network, 2023.
Available online with free registration. Last accessed October 25, 2024.
Management
Surgical Intervention: Risk-Reducing Gastrectomy
Risk-reducing total gastrectomy (RRTG) with esophagojejunostomy remains the treatment of choice for individuals with CDH1pathogenic variants.[
Functional outcomes after RRTG vary, but weight loss is universal. One series reported an average weight loss of 15% to 20% postsurgery.[
An international group of experts have published consensus guidelines for follow-up of patients with hereditary diffuse gastric cancer (HDGC) after RRTG.[
Endoscopic Surveillance
RRTG is generally considered the treatment of choice for patients with HDGC since it minimizes gastric cancer risk by removing high-risk gastric tissue (given the lack of consistently identifiable precursor lesions on endoscopy).[
Although RRTG remains the gold standard recommendation for HDGC, a multidisciplinary discussion on management options is favored based on the following: emerging evidence on endoscopic surveillance, the unpredictable rate of progression from intramucosal signet cells and T1a lesions to advanced gastric cancer, and morbidity of surgery. There is a role for endoscopic surveillance in HDGC, specifically for patients who decline surgery or choose to defer RRTG when they are diagnosed with HDGC. A prospective cohort study analyzed 270 asymptomatic CDH1 carriers (median age, 46.6 y; interquartile range [IQR], 36.5–59.8) who declined RRTG and participated in endoscopic surveillance with the Cambridge protocol (9%) or Bethesda protocol (91%).[
Other scenarios that may prompt endoscopic surveillance include the following:[
- Patients with family histories suggestive of HDGC but for whom no pathogenic variant has been identified.
- Patients with family histories of intestinal-type gastric cancer. This type of gastric cancer is usually not associated with CDH1 pathogenic variants. Therefore, endoscopic surveillance needs to be individualized for these patients.
- Patients who have a CDH1 pathogenic variant and choose to defer RRTG for a year or more. This includes patients who undergo genetic testing at a young age and test positive for a CDH1 pathogenic variant. Risk-reducing gastrectomy is generally not recommended in individuals who are younger than 18 to 20 years. Despite being warned about endoscopic biopsy's poor rate of HDGC precursor detection, many patients (especially those who are older) will only consider gastrectomy in the event of a positive biopsy.
There are no definitive, identifiable features of early diffuse gastric cancer or diffuse gastric cancer precursors that can be found on endoscopy. Targeted biopsies of ulcers, erythema, nodularity/nodules, polyps, erosions, and scars revealed SSRC in less than 10% of samples.[
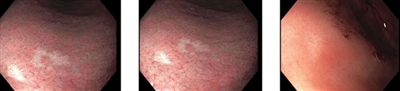
Figure 3. Several small, white, slightly depressed areas are best seen with narrow-band imaging. These lesions can resemble scars from previous endoscopic biopsies. However, the images above are from a patient who did not have a prior esophagogastroduodenoscopy. A microscopic signet ring cell carcinoma focus was found on this biopsy, although nontargeted biopsies in this patient were also positive for signet ring cell carcinoma.
Another study reported on 33 patients with CDH1 pathogenic variants (median age, 32 y) who were members of the original Māori family in which HDGC was first discovered. Participants underwent 99 surveillance endoscopies, 93 of which were performed with a mucolytic N-acetylcysteine followed by Congo red–methylene blue dye.[
If an endoscopic approach is taken for HDGC management, endoscopic surveillance is recommended annually unless signet ring cell features are found. If signet ring cell lesions are found on endoscopy, discussion with a multidisciplinary team is warranted.[
Breast Cancer Risk Management
In females with a CDH1 pathogenic variant, screening modalities for breast cancer include the following:
Breast magnetic resonance imaging (MRI):
- The National Comprehensive Cancer Network (NCCN) recommends consideration of breast MRI with contrast beginning at age 30 years.[
17 ] - The International Gastric Cancer Linkage Consortium (IGCLC) recommends that annual breast MRI begin at age 30 years since mammography is not effective at identifying lobular breast cancer.[
1 ,18 ]
Mammography:
- NCCN and IGCLC have differing recommendations regarding the age to begin mammography screening.
- NCCN recommends that annual mammography screenings begin at age 30 years.[
17 ] - IGCLC recommends that annual mammography screenings begin between the ages of 35 and 40 years, based on the patient's breast cancer risk factors, like breast density.[
1 ]
Other imaging:
- The IGCLC states that supplementary screening of dense breasts with ultrasound can be considered, especially when MRI is not available, contraindicated, or declined.[
1 ]
Bilateral risk-reducing mastectomy can be considered for women with a CDH1 pathogenic variant based on a strong family history of breast cancer, additional breast cancer risk factors, and/or personal choice.[
References:
- Blair VR, McLeod M, Carneiro F, et al.: Hereditary diffuse gastric cancer: updated clinical practice guidelines. Lancet Oncol 21 (8): e386-e397, 2020.
- Vos EL, Salo-Mullen EE, Tang LH, et al.: Indications for Total Gastrectomy in CDH1 Mutation Carriers and Outcomes of Risk-Reducing Minimally Invasive and Open Gastrectomies. JAMA Surg 155 (11): 1050-1057, 2020.
- Strong VE, Gholami S, Shah MA, et al.: Total Gastrectomy for Hereditary Diffuse Gastric Cancer at a Single Center: Postsurgical Outcomes in 41 Patients. Ann Surg 266 (6): 1006-1012, 2017.
- Stillman MD, Kusche N, Toledano S, et al.: Short and long-term outcomes of prophylactic total gastrectomy in 54 consecutive individuals with germline pathogenic mutations in the CDH1 gene. J Surg Oncol 126 (8): 1413-1422, 2022.
- Asif B, Sarvestani AL, Gamble LA, et al.: Cancer surveillance as an alternative to prophylactic total gastrectomy in hereditary diffuse gastric cancer: a prospective cohort study. Lancet Oncol 24 (4): 383-391, 2023.
- Charlton A, Blair V, Shaw D, et al.: Hereditary diffuse gastric cancer: predominance of multiple foci of signet ring cell carcinoma in distal stomach and transitional zone. Gut 53 (6): 814-20, 2004.
- van der Post RS, Gullo I, Oliveira C, et al.: Histopathological, Molecular, and Genetic Profile of Hereditary Diffuse Gastric Cancer: Current Knowledge and Challenges for the Future. Adv Exp Med Biol 908: 371-91, 2016.
- Corso G, Magnoni F, Nicastro V, et al.: Global distribution of prophylactic total gastrectomy in E-cadherin (CDH1) mutations. Semin Oncol 49 (2): 130-135, 2022.
- Bres C, Voron T, Benhaim L, et al.: Management of Pathogenic CDH1 Variant Carriers Within the FREGAT Network: A Multicentric Retrospective Study. Ann Surg 276 (5): 830-837, 2022.
- Davis JL, Ripley RT: Postgastrectomy Syndromes and Nutritional Considerations Following Gastric Surgery. Surg Clin North Am 97 (2): 277-293, 2017.
- Roberts G, Benusiglio PR, Bisseling T, et al.: International Delphi consensus guidelines for follow-up after prophylactic total gastrectomy: the Life after Prophylactic Total Gastrectomy (LAP-TG) study. Gastric Cancer 25 (6): 1094-1104, 2022.
- Norton JA, Ham CM, Van Dam J, et al.: CDH1 truncating mutations in the E-cadherin gene: an indication for total gastrectomy to treat hereditary diffuse gastric cancer. Ann Surg 245 (6): 873-9, 2007.
- Jacobs MF, Dust H, Koeppe E, et al.: Outcomes of Endoscopic Surveillance in Individuals With Genetic Predisposition to Hereditary Diffuse Gastric Cancer. Gastroenterology 157 (1): 87-96, 2019.
- Lee CYC, Olivier A, Honing J, et al.: Endoscopic surveillance with systematic random biopsy for the early diagnosis of hereditary diffuse gastric cancer: a prospective 16-year longitudinal cohort study. Lancet Oncol 24 (1): 107-116, 2023.
- Fujita H, Lennerz JK, Chung DC, et al.: Endoscopic surveillance of patients with hereditary diffuse gastric cancer: biopsy recommendations after topographic distribution of cancer foci in a series of 10 CDH1-mutated gastrectomies. Am J Surg Pathol 36 (11): 1709-17, 2012.
- Shaw D, Blair V, Framp A, et al.: Chromoendoscopic surveillance in hereditary diffuse gastric cancer: an alternative to prophylactic gastrectomy? Gut 54 (4): 461-8, 2005.
- National Comprehensive Cancer Network: NCCN Clinical Practice Guidelines in Oncology: Genetic/Familial High-Risk Assessment: Breast, Ovarian, and Pancreatic. Version 2.2024. Plymouth Meeting, Pa: National Comprehensive Cancer Network, 2023.
Available online with free registration. Last accessed September 18, 2024. - Pereslucha AM, Wenger DM, Morris MF, et al.: Invasive Lobular Carcinoma: A Review of Imaging Modalities with Special Focus on Pathology Concordance. Healthcare (Basel) 11 (5): , 2023.
Latest Updates to This Summary (10 / 25 / 2024)
The PDQ cancer information summaries are reviewed regularly and updated as new information becomes available. This section describes the latest changes made to this summary as of the date above.
Added
Revised
Revised
Revised
The
The
The
This summary is written and maintained by the
About This PDQ Summary
Purpose of This Summary
This PDQ cancer information summary for health professionals provides comprehensive, peer-reviewed, evidence-based information about the genetics of hereditary diffuse gastric cancer. It is intended as a resource to inform and assist clinicians in the care of their patients. It does not provide formal guidelines or recommendations for making health care decisions.
Reviewers and Updates
This summary is reviewed regularly and updated as necessary by the
Board members review recently published articles each month to determine whether an article should:
- be discussed at a meeting,
- be cited with text, or
- replace or update an existing article that is already cited.
Changes to the summaries are made through a consensus process in which Board members evaluate the strength of the evidence in the published articles and determine how the article should be included in the summary.
The lead reviewers for Hereditary Diffuse Gastric Cancer are:
- Doreen Agnese, MD (The Ohio State University)
- Ilana Cass, MD (Dartmouth-Hitchcock Medical Center)
- Lee-may Chen, MD (UCSF Helen Diller Family Comprehensive Cancer Center)
- Mary B. Daly, MD, PhD (Fox Chase Cancer Center)
- Grace-Ann O. Fasaye, ScM, CGC (National Cancer Institute)
- Gautam Mankaney, MD (Virginia Mason Franciscan Health)
- Tuya Pal, MD, FACMG, FCCMG (Vanderbilt-Ingram Cancer Center)
- Padma Sheila Rajagopal, MD, MPH, MSC (National Cancer Institute)
Any comments or questions about the summary content should be submitted to Cancer.gov through the NCI website's
Levels of Evidence
Some of the reference citations in this summary are accompanied by a level-of-evidence designation. These designations are intended to help readers assess the strength of the evidence supporting the use of specific interventions or approaches. The PDQ Cancer Genetics Editorial Board uses a
Permission to Use This Summary
PDQ is a registered trademark. Although the content of PDQ documents can be used freely as text, it cannot be identified as an NCI PDQ cancer information summary unless it is presented in its entirety and is regularly updated. However, an author would be permitted to write a sentence such as "NCI's PDQ cancer information summary about breast cancer prevention states the risks succinctly: [include excerpt from the summary]."
The preferred citation for this PDQ summary is:
PDQ® Cancer Genetics Editorial Board. PDQ Hereditary Diffuse Gastric Cancer. Bethesda, MD: National Cancer Institute. Updated <MM/DD/YYYY>. Available at:
Images in this summary are used with permission of the author(s), artist, and/or publisher for use within the PDQ summaries only. Permission to use images outside the context of PDQ information must be obtained from the owner(s) and cannot be granted by the National Cancer Institute. Information about using the illustrations in this summary, along with many other cancer-related images, is available in
Disclaimer
The information in these summaries should not be used as a basis for insurance reimbursement determinations. More information on insurance coverage is available on Cancer.gov on the
Contact Us
More information about contacting us or receiving help with the Cancer.gov website can be found on our
Last Revised: 2024-10-25
This information does not replace the advice of a doctor. Ignite Healthwise, LLC, disclaims any warranty or liability for your use of this information. Your use of this information means that you agree to the
Healthwise, Healthwise for every health decision, and the Healthwise logo are trademarks of Ignite Healthwise, LLC.
Page Footer
I want to...
Audiences
Secure Member Sites
The Cigna Group Information
Disclaimer
Individual and family medical and dental insurance plans are insured by Cigna Health and Life Insurance Company (CHLIC), Cigna HealthCare of Arizona, Inc., Cigna HealthCare of Illinois, Inc., Cigna HealthCare of Georgia, Inc., Cigna HealthCare of North Carolina, Inc., Cigna HealthCare of South Carolina, Inc., and Cigna HealthCare of Texas, Inc. Group health insurance and health benefit plans are insured or administered by CHLIC, Connecticut General Life Insurance Company (CGLIC), or their affiliates (see
All insurance policies and group benefit plans contain exclusions and limitations. For availability, costs and complete details of coverage, contact a licensed agent or Cigna sales representative. This website is not intended for residents of New Mexico.