General Information About Male Breast Cancer
Incidence and Mortality
Estimated new cases and deaths from breast cancer (men only) in the United States in 2025:[1]
- New cases: 2,800.
- Deaths: 510.
Male breast cancer is rare.[2] Fewer than 1% of all breast carcinomas occur in men.[3,4] The mean age at diagnosis is between 60 and 70 years; however, men of all ages can be affected by the disease.
Anatomy
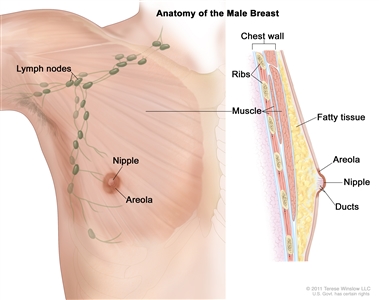
Anatomy of the male breast. The nipple and areola are shown on the outside of the breast. The lymph nodes, fatty tissue, ducts, and other parts of the inside of the breast are also shown.
Risk Factors
Predisposing risk factors for male breast cancer appear to include:[5,6]
- Radiation exposure to breast/chest.
- Estrogen use.
- Diseases associated with hyperestrogenism, such as cirrhosis or Klinefelter syndrome.
- Family health history: Definite familial tendencies are evident, with an increased incidence seen in men who have a number of female relatives with breast cancer.
- Major inheritance susceptibility: Increased male breast cancer risk has been reported in families with BRCA pathogenic variants, although risk appears to be higher with inherited BRCA2 variants than with BRCA1 variants.[7,8] At age 70 years, men have an estimated cumulative breast cancer risk of 1.2% if they have BRCA1 pathogenic variants and 6.8% if they have BRCA2 pathogenic variants.[9] Other genes may be involved in male breast cancer predisposition, including pathogenic variants in the PTEN tumor suppressor gene, TP53 (Li-Fraumeni syndrome), PALB2, and in mismatch repair genes associated with Lynch syndrome (also called hereditary nonpolyposis colorectal cancer).[10,11,12] For more information, see the sections on High-Penetrance Breast and/or Gynecologic Cancer Susceptibility Genes in Genetics of Breast and Gynecologic Cancers, and Male Breast Cancer Screening and Surveillance for BRCA1/2 Carriers in BRCA1 and BRCA2: Cancer Risks and Management.
Clinical Features
Most breast cancers in men present with a retroareolar mass. Other signs include:
- Nipple retraction.
- Bleeding from the nipple.
- Skin ulceration.
- Peau d'orange.
- Palpable axillary adenopathy.
Because of delays in diagnosis, breast cancer in men is more likely to present at an advanced stage.[2,5,13]
Diagnostic Evaluation
Breast imaging should be performed when breast cancer is suspected. The American College of Radiology recommends ultrasonography as the first imaging modality in men younger than 25 years because breast cancer is highly unlikely. Mammography is performed if ultrasonography findings are suspicious.
For men aged 25 years or older, or those who have a highly concerning physical examination, mammography is recommended as the initial test and ultrasonography is useful if mammography is inconclusive or suspicious.[14] Suspicious findings should be confirmed with a core biopsy. If the presence of tumor is confirmed, estrogen receptor, progesterone receptor, and human epidermal growth factor type 2 (HER2) expression/amplification should be evaluated.[15]
For more information, see the Diagnosis section in Breast Cancer Treatment.
Histopathologic Classification
Infiltrating ductal cancer is the most common tumor type of breast cancer in men, while invasive lobular carcinoma is very rare.[16] Breast cancer in men is almost always hormone receptor positive. In a male breast cancer series, 99% of the tumors were estrogen receptor positive, 82% were progesterone receptor positive, 9% were HER2 positive, and 0.3% were triple negative.[16]
Prognosis and Predictive Factors
Tumor size, lymph node involvement, and grade are anatomical prognostic factors, while estrogen receptor, progesterone receptor, and HER2 status are predictive of response to therapy.
A more advanced stage at diagnosis confers a worse prognosis for men with breast cancer.[2,5,13] A study found that mortality after breast cancer diagnosis was higher in male patients than in female patients. This disparity appeared to persist after accounting for clinical characteristics, treatment factors, and access to care, suggesting that biological factors and treatment efficacy may play a role.[17]
References:
- American Cancer Society: Cancer Facts and Figures 2025. American Cancer Society, 2025. Available online. Last accessed January 16, 2025.
- Giordano SH, Cohen DS, Buzdar AU, et al.: Breast carcinoma in men: a population-based study. Cancer 101 (1): 51-7, 2004.
- Borgen PI, Wong GY, Vlamis V, et al.: Current management of male breast cancer. A review of 104 cases. Ann Surg 215 (5): 451-7; discussion 457-9, 1992.
- Fentiman IS, Fourquet A, Hortobagyi GN: Male breast cancer. Lancet 367 (9510): 595-604, 2006.
- Giordano SH, Buzdar AU, Hortobagyi GN: Breast cancer in men. Ann Intern Med 137 (8): 678-87, 2002.
- Hultborn R, Hanson C, Köpf I, et al.: Prevalence of Klinefelter's syndrome in male breast cancer patients. Anticancer Res 17 (6D): 4293-7, 1997 Nov-Dec.
- Wooster R, Bignell G, Lancaster J, et al.: Identification of the breast cancer susceptibility gene BRCA2. Nature 378 (6559): 789-92, 1995 Dec 21-28.
- Thorlacius S, Tryggvadottir L, Olafsdottir GH, et al.: Linkage to BRCA2 region in hereditary male breast cancer. Lancet 346 (8974): 544-5, 1995.
- Tai YC, Domchek S, Parmigiani G, et al.: Breast cancer risk among male BRCA1 and BRCA2 mutation carriers. J Natl Cancer Inst 99 (23): 1811-4, 2007.
- Ding YC, Steele L, Kuan CJ, et al.: Mutations in BRCA2 and PALB2 in male breast cancer cases from the United States. Breast Cancer Res Treat 126 (3): 771-8, 2011.
- Silvestri V, Rizzolo P, Zanna I, et al.: PALB2 mutations in male breast cancer: a population-based study in Central Italy. Breast Cancer Res Treat 122 (1): 299-301, 2010.
- Boyd J, Rhei E, Federici MG, et al.: Male breast cancer in the hereditary nonpolyposis colorectal cancer syndrome. Breast Cancer Res Treat 53 (1): 87-91, 1999.
- Ravandi-Kashani F, Hayes TG: Male breast cancer: a review of the literature. Eur J Cancer 34 (9): 1341-7, 1998.
- Mainiero MB, Lourenco AP, Barke LD, et al.: ACR Appropriateness Criteria Evaluation of the Symptomatic Male Breast. J Am Coll Radiol 12 (7): 678-82, 2015.
- Giordano SH: A review of the diagnosis and management of male breast cancer. Oncologist 10 (7): 471-9, 2005.
- Cardoso F, Paluch-Shimon S, Senkus E, et al.: 5th ESO-ESMO international consensus guidelines for advanced breast cancer (ABC 5). Ann Oncol 31 (12): 1623-1649, 2020.
- Wang F, Shu X, Meszoely I, et al.: Overall Mortality After Diagnosis of Breast Cancer in Men vs Women. JAMA Oncol 5 (11): 1589-1596, 2019.
